Quickly and recently, collagen peptides have become one of the most sought-after products in the health and wellness aisle. A global wave of new product launches is fuelling the trend; but, with significant variations in source and dose, how to cut through the complexity?
Collagen is continuing to maintain its upward trajectory in 2019. However, ask ten different people what makes one collagen product better than the other and you will be given — at least — ten different answers. “The recent growth in collagen stems from a very broad group of consumers — the generational shift of physically active people who are driving the evolution of the sports nutrition market,” says Suzane Leser, Director of Nutrition Communication at GELITA AG.
Collagen and active nutrition consumers
The growth is attributed to the increasing awareness of the vital role that collagen-rich tissues, such as tendons, ligaments and muscle fascia, play in adapting to training and protecting against injuries. Adding to this, there is the unique appeal of collagen’s ability to reduce the age-related concept of whole-body decline.
For the active nutrition end of the sports supplement market, being in good shape is no longer just a body image issue; there’s the turning point when the body starts to show the first signs of physical decline and the wear and tear from previous decades.
The lifestyle impact is very real. Although soft tissue injuries account for more than 70% of the time away from competitive sports, there are also the injuries caused by running, cycling and fitness programmes that get in the way of adults who did not necessarily grow up being active, but are striving to transform their lifestyles and age well by adopting regular exercise.
A snapshot of the Europe/US collagen-based injury remedy market — performed by Nutrition Integrated, UK, in collaboration with GELITA — reveals a clear opportunity to develop the understanding of the category and to advance public knowledge about collagen.
“Collagen is a key trend with good scientific support and practical end user benefit applications. However, the growth of collagen products has given rise to a significant range of doses, sources and terminologies used to describe the source. As such, it isn’t easy to navigate. Just imagine if this could be improved, how much more scalable products would perform in the mainstream environment,” outlined Nick Morgan, Director at Nutrition Integrated.
Challenges such as these are nothing new for GELITA, the business that once revolutionised healthy ageing by successfully establishing its specific bioactive collagen peptide VERISOL in beauty-from-within products, merging both need-states and bringing a confident and positive emotional state to the healthy ageing category.
Need-state driven products
Nutrition Integrated revealed that, despite the need to be consumer centric, an estimated 73% of collagen products in the injury space are still ingredient led, emphasising that although collagen is on trend, continued differentiation is a task.
And, whereas many collagen solutions try to be all things to all people, GELITA offers a well-defined and substantiated portfolio of Bioactive Collagen Peptides (BCPs). Using product-specific science and communication, GELITA has succeeded in enabling customers to curate and personalise the best end-product solutions in markets such as beauty-from-within and joint health.
Bioactive collagens, hydrolysates and types: myth or reality?
Nutrition Integrated highlighted education as key to continued consumer traction. The sources and the terms currently used to describe collagen sources are not clear … and the wide range of doses is hard for consumers to rationalise. GELITA places a strong focus on BCPs, which is based on more than 150 years of manufacturing and researching gelatin and collagen proteins. GELITA has been able to identify, characterise and consistently obtain the different bioactive peptides that deliver the appropriate health benefits (Table I).
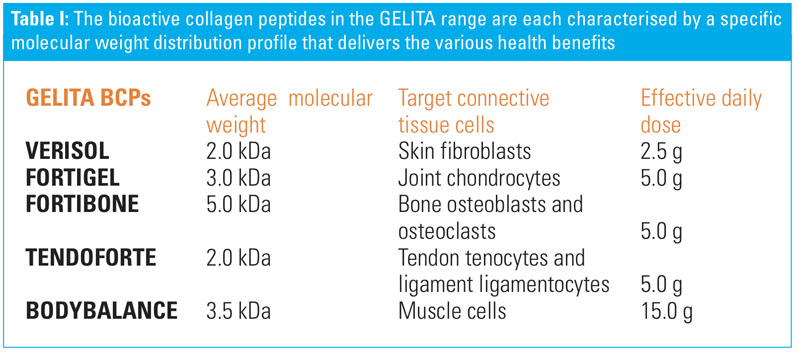
GELITA uses a special enzymatic hydrolysis process to produce its bioactive peptides from native collagen protein. The process is similar to human digestion, albeit highly specific, to consistently obtain the right sequences for the desired health benefit.
“Although gut digestion can break down collagen protein into peptides, the digestion is random and does not always yield the specific active sequences. Hence, if the use is for therapeutic or cosmetic purposes, it’s best to take collagen from supplemental bioactive peptides, rather than from food sources or generic collagen hydrolysates,” suggests Suzane.
In light of the diversity within the collagen peptides market, the following sections address the three biggest collagen myths a manufacturer may encounter when planning a strategic product portfolio approach in sports and active nutrition.
Myth #1: collagen is not the best protein source for sports nutrition
“Bioactive collagen peptides should not be in the protein bucket,” says Suzane. In basic nutrition, because collagen displays a low essential amino acid profile, it’s still often categorised as an incomplete protein source by the current methods used to assess protein quality. This is particularly challenging in sports nutrition wherein the primary role of protein is to provide all the essential amino acids for muscle recovery and growth.
However, Suzane adds: “The protein quality score of collagen does not apply to the effects of bioactive collagen peptides. The bioactive mode of action is beyond the basic nutritional role of protein in terms of contributing essential amino acids to meet daily needs.”
The positive impact of specific bioactive collagen peptides is delivered by a stimulatory effect on the target cells. It can be assumed that these bioactive peptides bind to specific receptors found on the surface of each type of cell. This stimulates the metabolism of the target cell to produce more extracellular matrix proteins, both in normal as well as healing collagen-rich tissues, such as bones, tendons and ligaments, skin and cartilage.
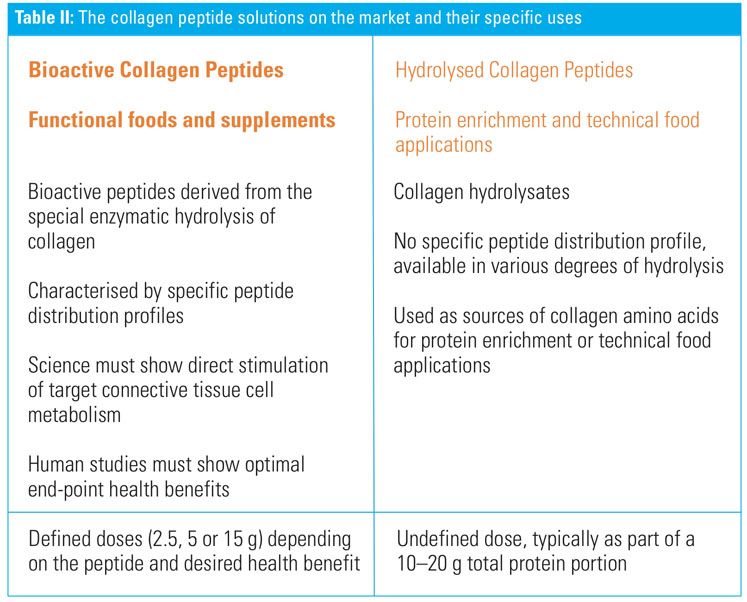
“It’s time to evolve protein thinking to a higher level. Two decades ago, the focus was on meeting basic needs for protein and essential amino acids to combat malnutrition and prolong life expectancies. Today, we are looking beyond basic needs, focusing on conditionally essential amino acids and on bioactive food compounds as regulators of metabolism for optimal and long-term health benefits ... and to address the specific physiological needs posed by ageing and exercise,” says Suzane (Table II).
Myth #2: joint health and the collagen superfamily
The individual collagen types have been labelled with Roman numerals in the order that they were discovered. Today, more than 28 types are known, which are grouped according to differences in their structural features as they appear in the body. On the shelf, the most commonly found types are I and II, which are the main structural collagens found in the body (type I being the most abundant and broadly distributed).
Type I makes up 90% of the body’s total collagen, providing a great deal of both mechanical and tensile strength to bones, ligaments, tendons and skin, as well as in fibrous cartilage. Type II is predominant in hyaline cartilage.
“The distribution of collagen types in the body is very complex. Regardless of where structural collagens appear in the body, as a nutritional source, the segmentation of collagen peptides by type is not at all important,” says Suzane. She adds: “The bioactivity of collagen peptides is independent of the native collagen types I, II or III; to obtain the bioactive peptide sequences, the process requires a specific hydrolytic step and denaturation of the original collagen type for the peptide to be effective.”
GELITA obtains its bioactive collagen peptides from a mixture of collagen types and, according to various preclinical trials, types I and II collagens have been shown to be nearly identical in terms of protein sequence (approx. 85%). The small difference has no influence on the bioactivity or cell stimulatory effect when broken down to the bioactive collagen peptides.
Myth #3: bioactive collagen peptides do not survive enzymatic digestion in the gut
Compared with all other proteins, collagen displays a unique amino acid profile that results in a rare peptide chain structure. This structure seems to facilitate the transport of the bioactive peptides across the gut wall. It was once believed that proteins must be broken down to amino acids or di- and tri-peptides before being absorbed by the healthy gut. However, there is now evidence that certain high molecular weight peptides, and even small proteins, can penetrate the gut barrier intact (to some extent).
Suzane explains: “It is speculated that peptides that contain high concentrations of the amino acid proline seem to be more resistant to gut hydrolysis; proline forms very strong peptide bonds that are more resistant to the action of digestive enzymes. What’s unique about collagen is that it’s remarkably rich in proline — which comprises 25% of the total amino acids in collagen peptides, a high proportion not seen in any other protein source.”
Another specific characteristic of collagen peptides is the rare, single helix structure.
This unique configuration might enhance the bioavailability of collagen peptides, compared with that of the normal alpha helix structure typically found in globular proteins, such as milk and eggs.
“A peptide chain is often seen as a linear string of linked amino acids. However, each amino acid has a slightly different shape that influences the final structure of the peptide. The rare structure of collagen peptides, formed by frequent proline-hydroxyproline-glycine repeats, is more extended and narrower than a normal alpha helix, so that it provides the favourable folding and stability that seems to facilitate gut absorption,” says Suzane.
She adds: “Paracellular transport seems to be the preferred route through which bioactive collagen peptides are absorbed intact through the gut wall, and this might be because of the narrower and stiffer shape of the collagen peptides, which looks like a drill or a needle, compared with the spring-like shape of globular proteins.”
“We estimate that approximately 10% of the bioactive collagen peptides stay intact during digestion and are available for the stimulation of connective tissue cell metabolism. The remaining 90% may be digested into amino acids, which are important products of peptide digestion. They are the building blocks of new connective tissue proteins, once the target cells have been directly stimulated by the bioactive peptide fractions,” concludes Suzane.
In summary
Collagen has come a long way in terms of consumer perception, becoming one of the main sources of functional peptides and positioned right at the centre of highly desirable consumer need-states in sports and active nutrition. Boasting nearly 20 years of product-specific research, GELITA has contributed to making collagen so much more than a solution for joint health and aesthetics.
The company has established bioactive collagen peptides as functional foods by showing a positive effect on skin elasticity, the recovery of lost cartilage tissue and reduced activity related to joint pain.
Recent studies now also show tendon and ligament benefits for daily activities and sports, increased lean body mass in young and elderly men and in premenopausal women, and increased bone mineral density in postmenopausal women. With the current market picture in mind, we must expect even more science and innovation in the future to further develop applications for bioactive collagen peptides.
Bibliography
- https://blog.euromonitor.com/what-is-active-nutrition-and-how-can-health-and-wellness-players-leverage-the-concept.
- R.D. Alcock, et al., Int. J. Sport Nutr. Exerc. Metab. 29, 265–272 (2019).
- 3. C. Paul, et al., Nutrients 11(5), E1079. doi: 10.3390/nu11051079 (2019).
- H.C. Siebert, et al., Protein Cell. 1(4), 393–405 (2010).
- E. Proksch, et al., Skin Pharmacol. Physiol. 27, 47–55 (2014).
- T.E. McAlindon, et al., Osteoarthr. Cartil. 19, 399–405 (2011).
- D. Zdzieblik, et al., Appl. Physiol. Nutr. Metab. 42(6), 588–595 (2017).
- K.L. Clark, et al., Curr. Med. Res. Opin. 24, 1485–1496 (2008).
- S.F.E. Praet, et al., Nutrients, 11(1), E76. doi: 10.3390/nu11010076 (2019).
- P. Dressler, et al., J. Sports Sci. Med. 17, 198–304 (2018).
- D. Zdzieblik, et al., Br. J. Nutr. 114, 1237–1245 (2015).
- P. Jendricke, et al., Nutrients 11(4), E892. doi: 10.3390/nu11040892 (2019).
- D. König, et al., Nutrients 10(1), E97. doi: 10.3390/nu10010097 (2018).




