Hypercholesterolemia is a growing problem in western countries, representing one of the major cardiovascular risk factors.
The 2019 guidelines on dyslipidemias of the European Society of Cardiology (ESC)1 recommends using statins up to the highest tolerated dose as a first-line therapy in cholesterol management.
Statins are effective at managing high plasma levels of cholesterol by inhibiting the 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, a key enzyme in the cholesterol biosynthetic pathway. However, HMG-CoA reductase diminishes the production of mevalonic acid, a precursor of endogenously synthesised CoQ10, therefore the intake of statins often results in lower CoQ10 levels in both serum and muscles. Up to 10-20% of patients on statin medications refer to myopathies and muscle pain, which is, in the majority of cases, related to CoQ10 deficiency. The oral supplementation of CoQ10 may be an effective approach to reducing such an effect. However, the bioavailability of CoQ10 after oral intake is limited and high dosages are often required in order to cause a meaningful therapeutic effect.
The clinical trial recently published in Drug Design, Development and Therapy Journal 2 aimed to assess the efficacy of a bio-enhanced liquid CoQ10 formulation (Q-Factor) in dyslipidemic patients with intolerance to statins.
Clinical Study
In a double-blind, randomised, placebo-controlled clinical trial, 60 Caucasian patients, whose LDL-cholesterol levels were not adequately controlled and who were intolerant to statin therapy, were enrolled. Subjects were considered intolerant if they showed a CPK greater than three to 10 times the upper limit of normal (ULN) and/or a rise in the value of transaminases greater than three to five times the ULN and/or the onset of clinical symptoms such as asthenia and myalgia.
Patients were treated for three months with a liquid CoQ10 formulation (Q-Factor) at a low dosage (100 mg/day) since previous studies showed that its bioavailability was about three times higher than other CoQ10-based products available on the market3,4. The primary outcome was the assessment of clinical symptoms and the perception of pain evaluated throughout validated questionnaires (Clinical Index Score and VAS). The secondary outcome was the variation in lipid profile and safety parameters.
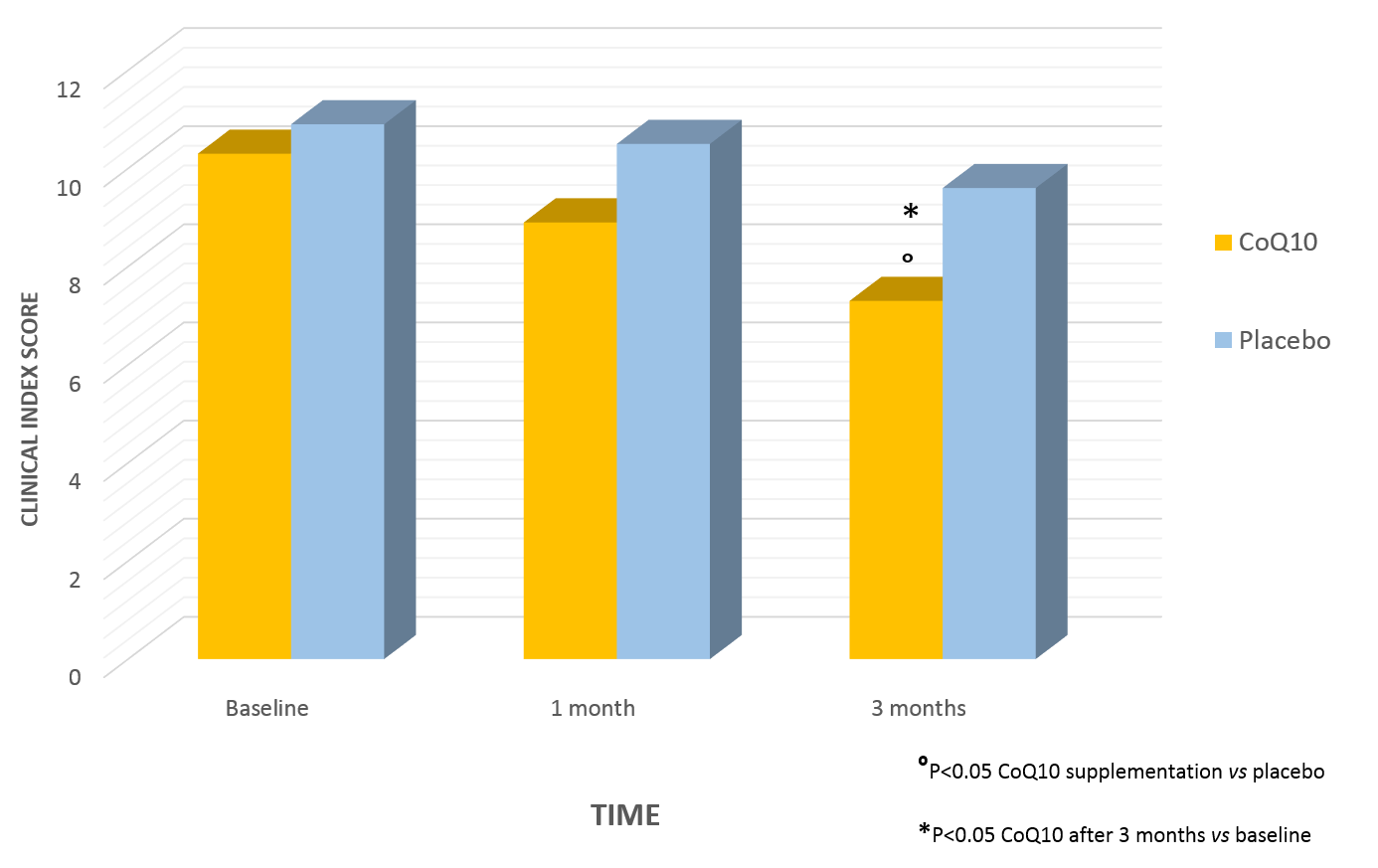
Study results
Coenzyme Q10 levels and Clinical Index Score were significantly improved after three months in the CoQ10 group, both compared to the baseline and the placebo group (°P<0.05 CoQ10 supplementation vs placebo; *P<0.05 CoQ10 after three months vs baseline).
After three months, patients treated with CoQ10 had lower total cholesterol (TC) and LDL-cholesterol compared to those taking the placebo (p<0.05 vs placebo).
Conclusion
The supplementation of Q-Factor in patients with previous intolerance to statins and on daily half-dose statin regimen, improves the perception of clinical symptoms such as asthenia, myalgia or pain. Q-Factor was safe and effective in preventing the worsening of the lipid profile that would be expected with a reduced dosage of statin.
Q-FACTOR in a nutshell
Q-FACTOR is a clinically tested dietary supplement that helps to maintain natural CoQ10 levels during a cholesterol-lowering statin therapy.

Q-FACTOR contains a patented water and fat soluble CoQ10 (Micro-sphere Dispersion Technology) that has been subject of several human bioavailability studies.
- Clinically proven
- Patented delivery system (Micro-sphere Dispersion Technology)
- Three times more easily absorbed than regular CoQ10
- 100% water and fat soluble
- On-the-go (liquid sachets)
- Available in drops
- Papaya flavoured
Visit Giellepi SpA SpA at CPhI, LIVE PHARMA CONNECT, Galleria level 1 stand G1A9.
References1. Mach et al., "ESC Scientific Document Group. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk," European Heart Journal, ehz455, https://doi.org/10.1093/eurheartj/ehz455.
2. G. Derosa, et al., "Coenzyme q10 liquid supplementation in dyslipidemic subjects with statin-related clinical symptoms: a double-blind, randomized, placebo-controlled study," Drug Design, Development and Therapy. 13:3647–3655 (2019).
3. RK. Chopra, et al., "Relative bioavailability of Coenzyme Q10 formulations in human subjects," Internat. J Nutr Res. 68:109-13 (1998).
4. MV. Miles, et al., "Bioequivalence of coenzyme Q10 from over-the-counter supplements," Nutrition Research, 919-29 (2002).




