Obesity is the fastest growing chronic disease — one that affects people of all ages. Its concomitant health risks are well documented, as is its role in the progression of other diseases, including cardiovascular disease, type 2 diabetes, respiratory problems and osteoarthritis.1
In the US, more than 67% of American adults are currently overweight or obese.2
Unfortunately, although obesity stems from consuming more calories than the body burns, more than 80% of overweight and obese individuals have trouble losing and maintaining weight loss through diet and exercise alone. These individuals recognise the serious health consequences of being overweight and often undertake drastic measures, including drug therapies and invasive surgeries, to achieve a healthier weight.
Non-invasive weight management solutions
Weight management treatments range from natural, oral solutions to risky, invasive surgical procedures. In the dietary supplement industry, new products constantly offer promises to dieters; yet, most are born from similar technology and mechanisms of action.
bmiSMART is the first weight management brand to introduce oral weight management treatments from InQpharm, a Zaluvida Group, which are based on continuous biotech innovations and research.
Current solutions include Litramine, which was awarded the first Class II medical device certification for a plant-based weight management product in the world, and Tanitol, the first enzyme inhibitor that achieved medical device certification by targeting multiple digestive enzymes.
Each bmiSMART product complex has been tested for safety and efficacy, including placebo-controlled, randomised human clinical trials published in peer-reviewed, indexed journals.3
A spectrum of weight management solutions
Weight management is complex and requires attention to numerous factors. Zaluvida Group has conducted more than 300 clinical studies on natural products and has developed a wealth of biotechnical health solutions, some of which have been commercialised by InQpharm North America (see Figure 1).
Weight loss through fat binding
InQpharm’s proprietary complex Litramine has its origins in Opuntia ficus-indica, a cactus also known as “prickly pear.” Through patented technologies, InQpharm has created a proprietary fibre complex with standardised lipophilic activity.
The resulting plant-based compound reduces dietary fat absorption through gastrointestinal fat binding, and has been shown to bind dietary fat at a level previously only achieved by prescription drugs, but without the undesirable side-effects.4
Litramine has undergone multiple human clinical trials, including a 14-week trial focused on weight loss and a 24-week weight maintenance study. Results on the 14-week weight loss study (2-week run-in period) are shown here (Figure 2).5
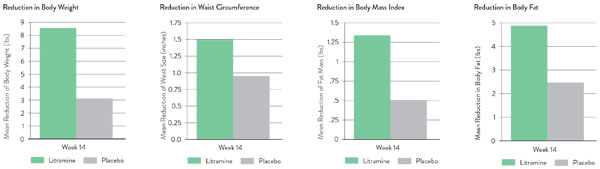
Figure 2: Litramine 14-week placebo-controlled, randomised, double-blind clinical trial results
Weight loss through digestive enzyme inhibition
InQpharm’s proprietary complex Tanitol is formulated with a unique blend of L-arabinose, a naturally occurring pentose, and grape marc extract. L-Arabinose works by inhibiting the hydrolysis of sugar (sucrose) to glucose and fructose by intestinal sucrase, thus reducing glucose absorption in the intestine.
Grape marc extract contains tannins that form complexes with carbohydrate-digesting enzymes, such as alpha-amylase and alpha-glucosidase, and the fat-digesting enzyme, lipase. This reduces the breakdown of complex carbohydrates, disaccharides and fats, which in turn reduces the absorption of carbohydrate-derived and fat-derived calories.
Results from a 14-week weight loss study using Tanitol to inhibit digestive enzyme function are shown in Figure 3 (2-week run-in period).6
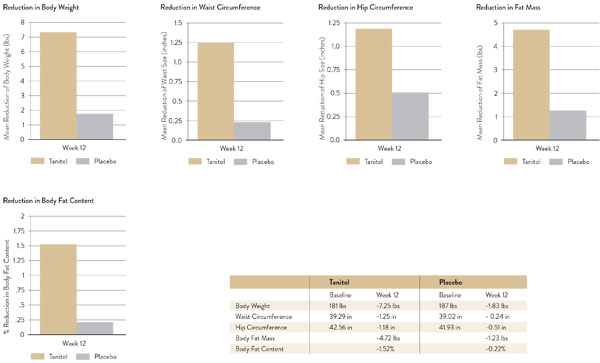
Figure 3: Tanitol 14-week placebo-controlled, randomised, double-blind clinical trial results
Weight loss through increased satiety
InQpharm’s Redusure is a patent-pending fibre complex based on glucomannan, carrageenan and xanthan gum. Designed to work like an impermanent, natural lap band, Redusure swells in the stomach, filling up to 43% of the volume, helping individuals to feel fuller for longer.
As food exits the stomach more slowly, the complex may delay the intestine’s absorption of glucose, helping to reduce the appetite and sustain feelings of satiety.
Redusure technology has been clinically studied and shown to help individuals lose weight significantly. In a 14-week clinical study (2-week run-in period) recently published in Advancement in Medical Plant Research, 95% of subjects consuming Redusure reported a moderate to strong feeling of satiety compared with only 59% for the placebo subjects. Additional study results are shown in Figure 4.7
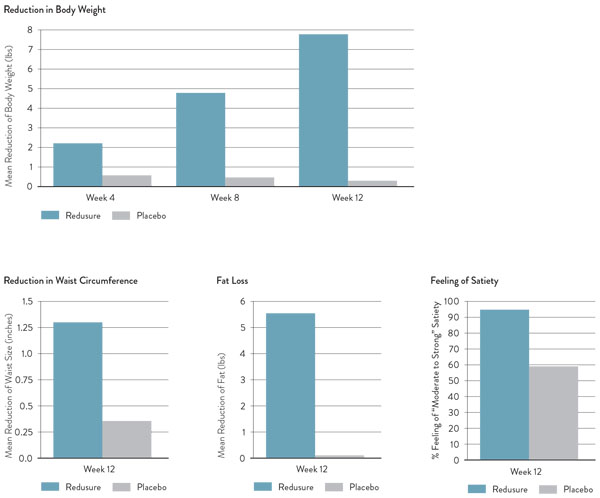
Figure 4: Redusure 14-week placebo-controlled, randomised, double-blind clinical trial results
As demonstrated by Zaluvida Group’s research and evidenced in InQpharm’s three bmiSMART products, efficacious, safe and easy to apply approaches to weight management do exist. They also allow adaption to individual dietary habits (such as preference for fatty foods or carbohydrate-rich diets) and can help to overcome initial hunger sensations while individuals are transitioning to a healthier diet.
References
1. www.who.int/nutrition/topics/obesity/en.
2. www.who.int/gho/ncd/risk_factors/overweight/en.
3. www.zaluvida.com.
4. R. Uebelhack, et al., “Effects of Cactus Fiber on the Excretion of Dietary Fat in Healthy Subjects: A Double Blind Randomized, Placebo-Controlled, Crossover Clinical Investigation,” Current Therapeutic Research 76, 39–44 (2014).
5. B. Grube, et al., “A Natural Fiber Complex Reduces Body Weight in the Overweight and Obese: A Double-Blind, Randomized, Placebo-Controlled Study,” Obesity 21, 58–64 (2013).
6. B. Grube, et al., “IQP-VV-102, A Novel Proprietary Composition for Weight Reduction: A Double-Blind Randomized Clinical Trial for Evaluation of Efficacy and Safety,” Evidence-Based Complementary and Alternative Medicine: http://dx.doi.org/10.1155/2015/413075 (2015).
7. U. Bongartz, et al., “Double-blind, Randomized, Placebo-Controlled, Bicentric Clinical Investigation to Evaluate the Benefit and Tolerability of Redusure IQP-AK-102 in Reducing Body Weight in Overweight and Obese Subjects,” Advancement in Medicinal Plant Research 4(3), 73–82 (2016).




