Natural Remedies, a global supplier of patented, clinically substantiated, innovative branded botanical ingredients, has announced the results of a new randomised double-blind clinical (RCT) study on its Turmacin, a naturally bioavailable extract of Turmeric. The clinical study, performed in healthy volunteers, significantly alleviated joint pain that arises upon exertion or exercise. The RCT was conducted at the St. Johns National Academy of Health Science, a premier research institute.
The study focused on 90 healthy volunteers who developed knee joint discomfort or pain of at least greater than 40 mm on VAS (Visual Analogue Scale) during 10 minutes of exercise on stepmill equipment. The volunteers were randomly assigned to consume placebo or Turmacin (500 mg) or Turmacin (1000 mg) daily for 12 weeks.
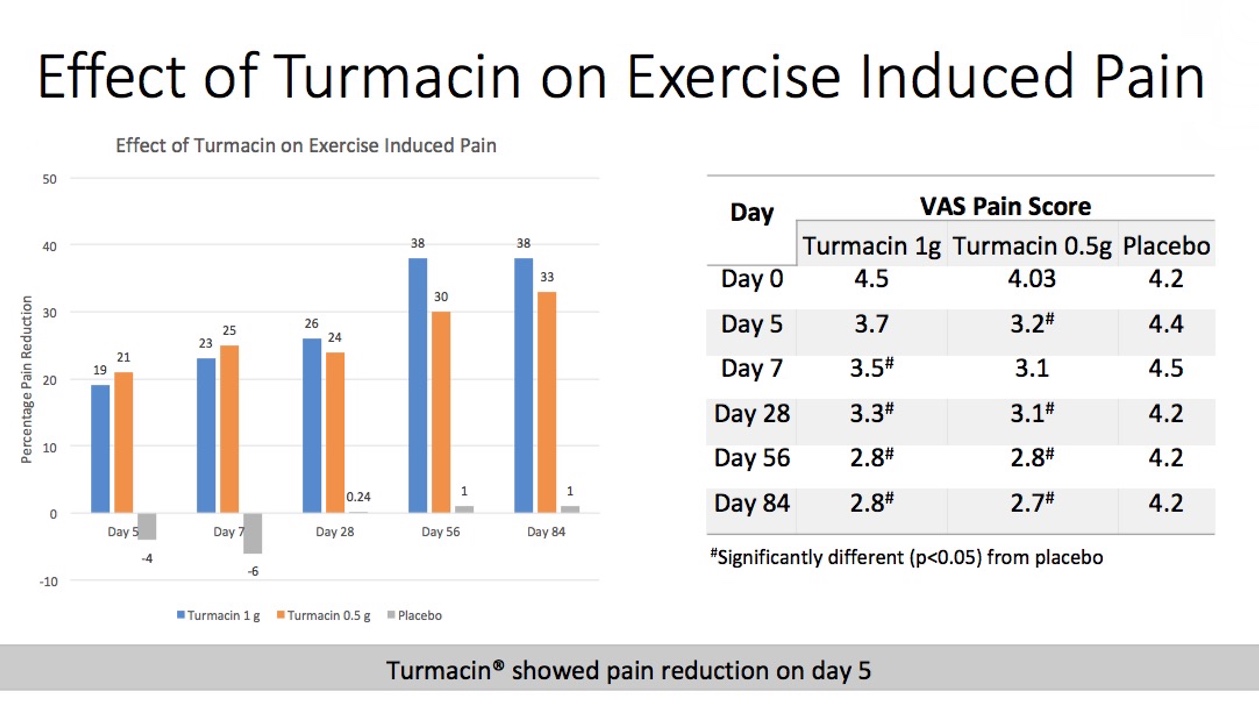
In the study, post-stepmill exercise pain scores (VAS 0-10) and the time of the initial pain/discomfort onset were recorded at baseline, and days 5, 7, 28, 56 and 84. Additionally, joint range of motion, and peak torque for muscle contraction at the joint were assessed at the end of 12 weeks using digital goniometry and isokinetic dynamometry respectively.
Sports recovery product applications with Turmacin now range from capsules and tablets, to shots, chews, gummies, protein powders, bars and beverages
According to the study authors, findings revealed that Turmacin significantly decreased the final pain scores from the first follow up of day 5 as compared to placebo group. A maximum decrease of 1.7 units on the VAS scale from baseline was seen in the Turmacin group on days 56 and 84; this is considered a clinically important difference.
In addition, the researchers observed Turmacin's efficacy in improving joint range of motion and preserving muscle function during relative physical inactivity when compared to placebo group at the end of study.
"Overall, the study suggests that Turmacin showed promising results for alleviating joint discomfort that arises during exertion or exercise," asserted Suresh Lakshmikanthan, global head of human health business for Natural Remedies. "Sports recovery product applications with Turmacin now range from capsules and tablets, to shots, chews, gummies, protein powders, bars and beverages."
Lakshmikanthan added that this is the second clinical trial on Turmacin substantiating its efficacy for joint and cartilage health. Turmacin, which was recently GRAS notified, is the first clinically tested naturally bioavailable composition derived from turmeric completely untouched by solvents and is standardised to contain bioactive polysaccharides Turmerosaccharides and does not contain curcuminoids. "This naturally bioavailable and water-soluble property of Turmacin, makes it an ideal choice for beverages and other functional food manufacturers," he said.
This study will be discussed in detail at SupplySide West. The presentation, "Water soluble components of turmeric for joint health," presented by Dr Deepak Mundkinajeddu, PhD, head of R&D for Natural Remedies, will take place on 17 October, from 1:30 to 1:50 pm, at Booth 1266.
As an industry leader, Natural Remedies contributed to improving the quality of Indian botanicals with active participation in the preparation of analytical monographs for various international pharmacopoeias. Natural Remedies is also a leading manufacturer and supplier of phytochemical reference standards from Indian medicinal plants, which are being used for quality and research purposes globally. Natural Remedies' human health care products provide quality combined with perfected expertise while offering global clients a one-stop-shop fulfilling all their requirements of quality health supplements and formulation ingredients.
US-based AIDP is the exclusive distributor of Turmacin in the US.
