To learn more about this approach, we spoke with the experts at Naturmega about the science behind phospholipid-bonded omega-3 and its potential implications for the category. As the omega-3 market continues to evolve, innovation is increasingly focused not only on the amount of EPA and DHA delivered, but also on how efficiently these fatty acids are absorbed and utilised by the body.
In this Q&A, the team behind Ruby-O® BPL-Omega-3 explains how advances in molecular lipid architecture may influence delivery efficiency, biomarker outcomes and product differentiation. They also discuss the findings from recent clinical research and what these developments could mean for the next generation of omega-3 nutraceutical formulations.
1. Why is molecular architecture becoming an increasingly important differentiator in the omega-3 category?
For many years, innovation in the omega-3 category focused primarily on dose and concentration, essentially how many milligrams of EPA and DHA a product could deliver.
Today, scientific understanding is shifting this perspective. Research increasingly shows that how omega-3 is delivered may be just as important as how much is consumed.
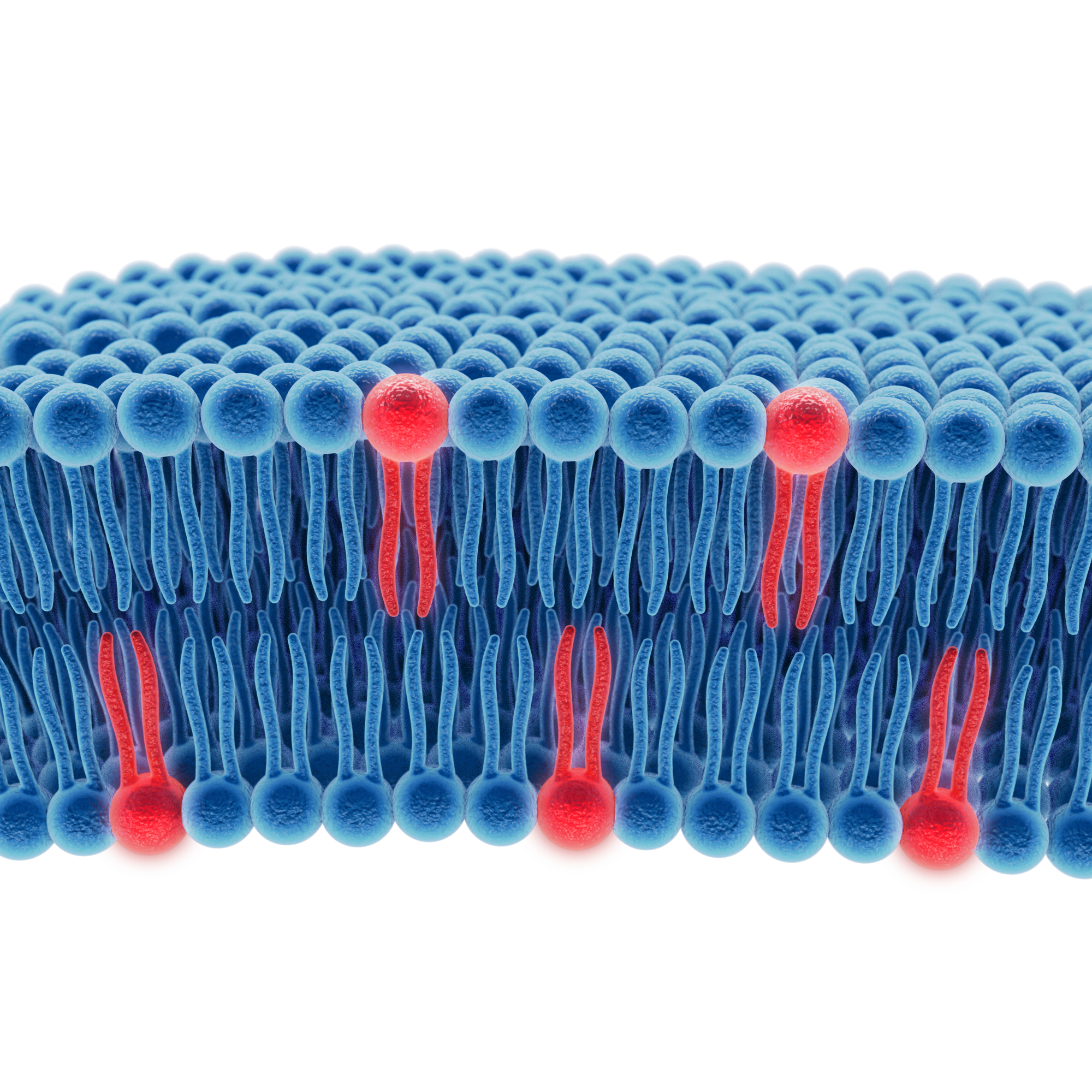
Omega-3 fatty acids exert many of their biological effects within cellular membranes, meaning that the molecular structure carrying them can play a critical role in how efficiently they are absorbed, transported and incorporated into tissues.
As the category matures, the conversation is gradually evolving from a simple question of “How much omega-3?” to a more nuanced one: “How efficiently is omega-3 delivered to where it actually works?”
This shift is positioning molecular lipid architecture as an emerging frontier for innovation in omega-3 nutrition, as researchers and ingredient developers explore new ways to optimise delivery and metabolic impact.
That shift is making molecular lipid architecture a new frontier for innovation in omega-3 nutrition.
2. How does phospholipid-bonded omega-3 differ from conventional fish oil formats?
Traditional omega-3 supplements typically deliver EPA and DHA in triglyceride (TG) or ethyl ester (EE) forms, which are considered neutral lipid structures.
Before these fatty acids can exert many of their biological functions, they must undergo several metabolic steps in the body. These neutral lipids are first digested and transported before the fatty acids are eventually incorporated into membrane phospholipids, where many of omega-3’s biological effects occur.
Phospholipid-bonded omega-3 approaches the challenge differently.
In this architecture, the omega-3 fatty acids are already linked to phospholipid molecules, which are the fundamental building blocks of cellular membranes and key components of lipid transport systems such as lipoproteins.
By delivering omega-3 in a structure that more closely resembles the body’s natural lipid organisation, this approach may facilitate more direct integration into biological membranes and lipid transport pathways, potentially improving delivery efficiency compared with conventional formats.
This biomimetic design reflects a growing focus in lipid science on optimising molecular structure to enhance metabolic utilisation.

3. What were the most important outcomes of the Ruby-O clinical study?
One of the most meaningful findings of the 12-week randomised clinical trial evaluating Ruby-O® BPL-O3™ was not only the average reduction in triglyceride levels, but the proportion of participants who were able to move out of established cardiovascular risk ranges.
The study showed that a significantly higher percentage of participants receiving Ruby-O achieved clinically relevant triglyceride thresholds compared with the conventional omega-3 control group.
For example, 50.0% of participants receiving Ruby-O reached triglyceride levels of ≤166 mg/dL, compared with 18.2% in the control group (p = 0.026).
Similarly, 40.9% of Ruby-O participants achieved ≤156 mg/dL, versus 13.6% in the control group (p = 0.041).
Most notably, 36.4% of individuals in the Ruby-O group achieved the clinically recognised target of ≤150 mg/dL, compared with 13.6% in the control omega-3 group, representing nearly a three-fold difference in reaching the clinical goal.
Another important outcome related to delivery efficiency. Despite providing a lower daily omega-3 dose, Ruby-O produced a greater increase in Omega-3 Index levels, an important biomarker associated with cardiovascular health.
These findings suggest that the molecular architecture through which omega-3 is delivered can play a significant role in determining biological outcomes, reinforcing the growing importance of delivery efficiency in omega-3 nutrition.
4. The study showed meaningful biomarker improvements at a lower dose. What does that suggest?
It highlights the importance of delivery efficiency in omega-3 nutrition.
Phospholipids are not simply carriers of fatty acids, they are structural components of cell membranes and key participants in lipid transport systems in the body.
When EPA and DHA are incorporated into phospholipids such as phosphatidylcholine (PC) or lysophosphatidylcholine (LPC), they may integrate more directly into physiological lipid pathways. In other words, the body may be able to recognise, transport and use these molecules more efficiently.
This can translate into greater biological impact per milligram of omega-3 consumed, which is consistent with the biomarker improvements observed in the clinical study.
For nutraceutical innovation, this reinforces an emerging concept in the category: it is not only the amount of omega-3 that matters, but how effectively it is delivered and utilised by the body.
5. Why is movement out of cardiovascular risk ranges clinically important?
Healthcare professionals often evaluate nutritional or therapeutic interventions based on whether they help patients move from one clinical risk category to another, not just on average biomarker reductions.
For example, lowering triglycerides below 150 mg/dL moves an individual from an elevated range into the normal category.
This type of categorical improvement is often considered more clinically meaningful than a modest average reduction, because it reflects a tangible improvement in cardiometabolic status.
For clinicians, reaching these thresholds can indicate:
• successful target attainment
• improved cardiometabolic health status
• potentially reduced need for additional therapeutic interventions
For consumers, achieving a defined health target also provides a clear and motivating signal of progress, which can reinforce long-term adherence to nutrition and lifestyle strategies.
6. What does “bonded phospholipid omega-3” mean in practical terms?
In practical terms, it means that EPA and DHA are structurally incorporated into the phospholipid fraction of the lipid matrix, rather than existing only in neutral lipid forms such as triglycerides.
Because phospholipids are naturally present in biological membranes and lipid transport particles, delivering omega-3 in this structure may help the fatty acids integrate more directly into physiological pathways.
To verify this architecture analytically, Naturmega developed a multi-technique characterisation approach, combining several complementary methods:
• column chromatography to separate lipid classes
• gas chromatography (GC) to quantify fatty acid composition
• HPLC to characterise phospholipid species
• NMR spectroscopy to provide structural confirmation
Together, these analytical tools allow researchers to confirm not only the total amount of EPA and DHA present, but also how those fatty acids are structurally organised within the lipid matrix, which is critical for understanding how the ingredient functions biologically.
7. How does molecular architecture influence variability in omega-3 responses?
One of the well-known challenges in omega-3 research is the high variability in clinical responses between individuals.
Factors such as genetics, metabolic status, diet and baseline lipid levels can all influence how people respond to omega-3 supplementation.
Improving the efficiency of molecular delivery may help reduce part of this variability.
By delivering omega-3 in a phospholipid-bonded, biomimetic structure, Ruby-O may facilitate more consistent incorporation of EPA and DHA into cellular membranes.
For this reason, the clinical study placed strong emphasis on responder analysis and target attainment, rather than relying solely on average biomarker changes. This approach provides a clearer picture of how many individuals achieve meaningful physiological improvements.
8. How can nutraceutical brands translate these findings into differentiated products?
The omega-3 category has become highly competitive, with many products offering similar claims around purity, sustainability and concentration.
Scientific advances in lipid delivery systems allow brands to move beyond commodity positioning and create products with stronger scientific narratives.
Instead of focusing only on higher EPA and DHA doses, brands can develop formulations built around:
• molecular lipid design
• delivery efficiency
• clinically meaningful outcomes
This represents a shift toward what many in the industry are beginning to describe as next-generation omega-3 formulations, where the emphasis moves from simply delivering more omega-3 to delivering omega-3 more intelligently.
9. Which product categories may benefit most from phospholipid-based omega-3?
The most immediate application is in cardiometabolic health, particularly for individuals with moderately elevated triglycerides who are looking for nutritional strategies to improve lipid markers.
However, the potential relevance of phospholipid-based omega-3 extends beyond cardiovascular health.
Because phospholipids are fundamental components of cell membranes and lipid transport systems, this delivery approach may also be relevant in areas such as:
• brain and cognitive health
• metabolic health
• healthy ageing
• premium omega-3 formulations
There is also increasing scientific interest in phospholipid and lysophospholipid forms of DHA, as research suggests they may facilitate more efficient transport to the brain compared with conventional triglyceride forms.
10. Why is clinically validated differentiation becoming essential in omega-3?
The omega-3 market has matured significantly over the past two decades.
Most products today already emphasise purity, sustainability and high EPA/DHA concentrations, which means that many offerings appear similar to consumers and healthcare professionals.
What increasingly differentiates ingredients is clinical evidence demonstrating meaningful health outcomes or improved delivery mechanisms.
Clinically studied delivery platforms allow brands to build stronger credibility with:
• healthcare professionals
• retailers and practitioners
• science-oriented consumers
Looking ahead, many experts believe the next phase of innovation in omega-3 will be driven by scientifically validated lipid architectures designed to improve biological delivery efficiency, rather than simply increasing omega-3 concentrations.

