One critical factor responsible for a tablet meeting acceptable quality attributes is its final weight.
The tablet weight is not only related to the active ingredient’s dosage, but it also directly affects the applied compression force and, subsequently, the resulting tablet strength, friability, disintegration and dissolution times.
Key considerations
Multiple steps and variables must be considered before the actual powder is filled into the die cavity, the most important of which are detailed below.
The hopper: Powder is initially loaded into the hopper of a tablet press and must have adequate flow properties to successfully enter the gravity or paddle feeder (also referred to as a force feeder).
Typical powder flow issues found in the hopper are ratholing, bridging and segregation. The hopper design can play a role in this as those with sharp angles and rough surfaces can inhibit powder flow.

Robert Sedlock
Hoppers designed for mass flow with high-quality surface finishes can address these issues. Understanding the rheological properties of the powder allows for the proper hopper design and a significant reduction of powder flow issues.
Vibration from the tablet press can cause powder particle size segregation — with larger particles percolating upward.
Products that require a high tonnage compression force may cause excessive vibration derived from punch/roller interactions; however, quality tablet presses that are designed for high compression forces will absorb the impact shock and may help to remediate segregation issues.
The amount of powder in the hopper will determine the head pressure of the powder going into the feeder, which can lead to tablet weight variability. A low-level proximity probe installed on the hopper is often used to alert the operator that the hopper is almost empty.
However, consideration in terms of positioning that sensor so that the material charge is always above a certain level to maintain the required head pressure is warranted.
The feeder: The feeder is the final component that delivers the powder to the die cavity. Common feeder designs can be gravity or force fed with single of multiple paddles.
Many variables are associated with feeders, including size, clearance above the die table, paddle design and speed. The speed of the paddles in the feeder is determined by the speed of the turret, number of punches installed on the turret and powder flow characteristics.
The purpose of the paddles is to maintain a constant charge of powder in the feeder for proper dosing into the die cavity.
Tablet press operators must be vigilant to avoid running the paddles at an excessive speed, which can cause issues with material packing and cause flow restrictions, overblending of the formulation components and/or particle segregation.
When dealing with highly compactible materials, packing may cause particle adhesion and prohibit the transfer of powder into the die.
Overblending is an issue when higher levels of lubricant (magnesium stearate) are used in the formulation and excessive paddle speeds cause the lubricant to coat a higher percentage of the particle surfaces.
This overblending can cause tablet quality issues such as lower tablet strengths, capping and extended disintegration/dissolution times.
This issue is exacerbated when powders are recycled from the fill cam/overfill, which is explained in more detail later. The correct feeder speed is the slowest speed that maintains an acceptable — and consistent — final tablet weight.
Fill cam selection: The fill cam is a lower cam located under the feeder and deliberately overfills the die cavity. Following the fill cam is the dosing cam, which will push out the excess material to achieve the target weight (see Figure 1).
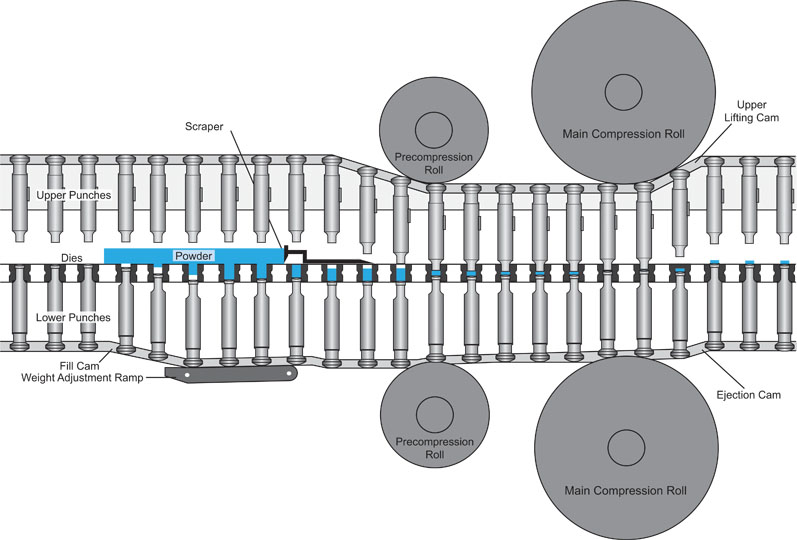
Figure 1: The fill cam and dosing cam of a rotary tablet press
The excess powder is removed by a scraper bar that directs the powder to a waste area or recycling path back into the feeder.
This two-step filling process is important. As the overfilled material is pushed up from the dosing cam and scraped off, the powder is consolidated and will help to ensure that the correct dosage reaches the die cavity.
By contrast, excessive filling and scraping will cause issues with either low material yields or excessive recycling and potential overblending as described earlier.
It is important to know that the proper fill cam size should be selected based on the dosing cam position. The recommended amount of overfill is 2–4 mm.
The fill cam size can be approximated by calculating the dosing cam depth to achieve the target weight and simply adding 2–4 mm. The dosing cam dimensions are based on the tooling geometry, material bulk density and tablet weight.
First, calculate the volume of powder to be compressed by dividing the final tablet weight by the bulk density of the material.
Then, divide the volume of powder to be compressed by the punch tip’s cross-sectional area, which is typically included on the tool drawing or can be provided by the supplier.
Powder characteristics
The above discussion focused on the equipment aspects of providing the tools to properly fill the die cavity. The powder blend and particle characteristics also play a major role in the die-filling process.
If powder simply doesn’t flow, the resulting tablet is affected. Some factors that should be considered to enhance flow are flow-aiding excipients such as glidants (talc or silicon dioxide) and granulation techniques including dry and wet processes.
Candidate formulations should be evaluated for their flow characteristics and performance under typical tablet press conditions.
The best method available for predicting powder performance throughout the tablet-making process is a measurement of powder rheology utilising one of the new instruments available on the market.
Conclusion
Understanding the powder die filling process is a crucial aspect of providing a batch of tablets that meets the expectations of patients and regulatory authorities. Powder and equipment engineering are key factors in the success of high-speed tablet manufacturing.




