Today’s consumer faces a vast amount of choice in the health and nutrition market, and this wide array of selection has raised the bar regarding what it takes for a new product to make it in this competitive sector. At the same time, product developers are demanding high quality, reliable and efficient options to help them create pharmaceutical solutions and nutritional supplements that they can be proud to attach their brand to. The needs of both can be met with Vcaps Plus capsules, produced from the plant-based hypromellose polymer (HPMC).
Unlike early generations of HPMC capsules, Vcaps Plus capsules are produced without any secondary gelling agents, which have been proven to delay dissolution under some circumstances and also lead to unwanted issues during product development. Not only do these pure, visually appealing capsules meet the wants and needs of the European consumer — in particular, high effectiveness and the peace of mind derived from a pure, organic product — but they have also demonstrated in numerous studies the superior performance that health and nutrition manufacturers demand. Vcaps Plus capsules offer numerous, clear product advantages: superior disintegration and dissolution; optimal product stability; reduced development times owing to ease of integration into existing production lines; and readiness for tough regulatory environments around the globe.
The Customer is Always Right
The health and nutrition market continues to expand and develop around the world, with emerging new major markets such as China and Brazil catching the industry’s attention. A 2012 Research and Markets report on the global nutraceutical market forecasts steady growth in this sector between 2012 and 2017, predicting potential in areas including India, the Middle East and Eastern Europe.1
As consumers of these products become increasingly savvy and selective about the products they choose, Vcaps Plus deliver the key properties that they seek. Because supplements are all about health, wellness and taking care of yourself, consumers want a product that matches their high standards: authenticity purity, and reliability. Non-GMO, pesticide-free, preservative-free and gluten-free, Vcaps Plus are created from a plant-based cellulose that enables them to be labelled as vegetarian, Kosher and Halal. Beautiful branding, colour and lustre options give manufacturers plenty of creative room to design a visually appealing product, with a smooth, high-end texture that ensures easy swallowing.
An Organic Edge
Vcaps Plus also offer a perfect solution for marketers looking to encapsulate organic products in Europe, creating an end-product that can be labelled as organic. Figures from around the globe clearly show that organic is booming — and organic designation is a clear asset to health and nutrition product marketers.
Consumers have become sophisticated with their environmental awareness and product choices
A Datamonitor report titled “The Next Step in the Ethical Consumerism Revolution” found that consumers have become sophisticated with their environmental awareness and product choices, which means that it’s no longer sufficient to market a product as “natural”. A 2008 study from the French marketing firm Expansion Consulteam came to the same conclusion, finding that consumers are largely willing to pay higher prices for a product with an official organic label. “Certification is synonymous with security. The organic label is the essential vector of added value.”
HPMC capsules such as Capsugel’s Vcaps Plus have been approved by the EU as an encapsulation material for organic products since December 2007, making them the perfect choice for organic supplement marketers seeking to take full advantage of today’s exploding market for organic health products.
The HPMC Advantage
The numerous advantages of Capsugel’s Vcaps Plus line stem from the benefits of HPMC as a base material. This plant-derived cellulose has been an option for vegetarian capsules in pharmaceutical applications since 1998.2 It is a physically stable polymer that’s resistant to stringent heat and humidity conditions, with a low moisture content that makes it suitable for moisture-sensitive ingredients.
Because the role of the capsule is to protect its contents from degradation or product changes, which means insulating against temperature fluctuations or moisture exposure, the stability of the capsule material is a critical factor in product development. Numerous in-house Capsugel and external studies have demonstrated the exemplary performance of HPMC in terms of both temperature and long-term storage stability.
The stability of the capsule material is a critical factor in product development
In fact, a Wyeth study found that empty Vcaps Plus capsules stored for one week at room temperature under different RH conditions had a three-fold lower average moisture content than gelatin capsules, and were less hygroscopic than gelatin as a whole, potentially reducing the risk of moisture transfer from the capsule to its contents.3 When exposed to low temperatures, Vcaps Plus did not display any changes in colour, transparency or disintegration; when exposed to high temperatures, they displayed greater resistance and less discolouration than hard gelatin capsules.
To Gel or Not to Gel?
Although many manufacturers have used secondary gelling agents to generate quick-setting HPMC capsules, these gelling systems may affect the dissolution of the capsule, causing undesirable delays in drug or supplement release. Capsugel’s own scientists set about developing an HPMC capsule free from gelling agents by assessing the impact of the mould pin temperature and conditions to achieve optimal capsule drying. The outcome of these studies was a thermal gelling process that eliminated the need to use a gelling agent and salts as cogelling agents, and the resulting product is the Vcaps Plus capsule.
The manufacturing process, in accordance with cGMP guidelines, was carefully defined to ensure the highest levels of capsule performance. The elastic yet robust polymer structure of the HPMC gives it a high machinability, and capsules were tested to ensure that factors of weight, dimensions, consistency and performance on filling machines remained of the highest standards. The HPMC surface remains smooth to facilitate easy capsule handling, and the option of either transparent or coloured opaque capsules offers flexible dosage formats.
Faster Dissolution, More Stability
For a health or nutrition supplement to deliver results, the disintegration and dissolution rates are of critical importance. For products that rely on dependable rapid and consistent dissolution, such as amino acid-based sport supplements or energy boosters, the Vcaps Plus advantage is clear. In October 2012, Capsugel’s Research and Development department presented key findings at the annual meeting of the American Association of Pharmaceutical Sciences, demonstrating that HPMC capsules without gelling agents, such as Vcaps Plus, deliver faster, consistent and more predictable dissolution than those containing gelling agents; that is, the high performance of gelatin with the stability advantages of the HPMC polymer.
HPMC capsules without gelling agents, such as Vcaps Plus, deliver faster, consistent and more predictable dissolution than those containing gelling agents
For the study, researchers measured the in vitro dissolution of HPMC hard capsules both with and without gelling agents in a variety of substances, including simulated milk fluid and liquids at pH 1.2 and pH 6.8. Whereas the capsules containing gelling agents were significantly affected by small changes in the pH or ionic properties of the liquids, the Vcaps Plus exhibited no changes in their steady dissolution rates. In one test, for example, Vcaps Plus capsules filled with caffeine had released more than 90% of their caffeine contents after 30 minutes in the test liquid, whereas the gelled capsules had released only 50% of their contents. In the simulated milk fluid, the difference was even more extreme: the Vcaps Plus capsules once again released more than 90% of their contents after 30 minutes, whereas the gelled capsules released less than 30%. Similar in-house tests also demonstrated that these superior dissolution rates continued regardless of capsule size (Figure 1).4
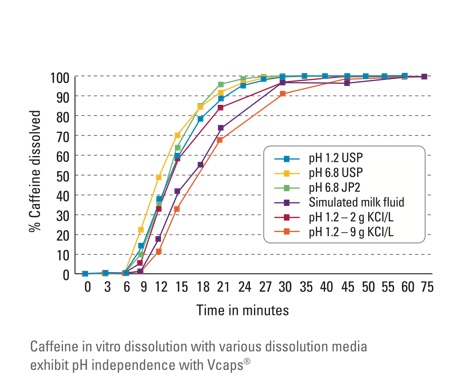
Figure 1: In vitro dissolution of caffeine in Vcaps Plus capsules
Because Vcaps Plus capsules demonstrate a consistent and pH-independent release of their contents, they deliver optimal product performance, an advantage pharmaceutical and nutraceutical manufacturers shouldn’t ignore.
From Conception to Market
While Vcaps Plus capsules set themselves apart with their superior dissolution rates, the capsules are nonetheless easily integrated into existing capsule filling, sealing and coating operations. Performance trials on many common high-speed capsule-filling machines indicate that Vcaps Plus capsules, with their smooth and shiny finish, show similar performance rates to gelatin capsules. Furthermore, Vcaps Plus capsules are also fully compatible with Capsugel’s hard capsule Fusion process, which produces one-piece, hermetically sealed, liquid-filled capsules. The global supply capability of the material eases scale-up to commercial production, making Vcaps Plus capsules the clear choice for companies ready to quickly introduce a new product to the market. In addition, all primary components of Vcaps Plus capsules are accepted for pharmaceutical and dietary supplement use in the major markets of the United States, Canada, Japan, Australia and the European Union.

Consumers want a product that matches their high standards: authenticity purity, and reliability
In summary, Vcaps Plus, developed without any secondary gelling agents, is an HPMC capsule that has all the physical and operational features that the pharmaceutical and nutraceutical industries and end-consumers are looking for. The HPMC material has been proven to be less hygroscopic and more temperature- and storage-stabile than gelatin, and also has the added bonus of being vegetarian. Being manufactured without any secondary gelling agents gives Vcaps Plus a significant performance edge when it comes to disintegration and dissolution, allowing for quick and consistent release of the capsule contents. Thanks to their compatibility with many common capsule filling machines and superior filling rates to traditional HPMC capsules, they can potentially reduce product development times as well. Pure, reliable, and high performing, Vcaps Plus capsules truly represent the next generation of capsule dosage forms.
References
1. Transparency Market Research, “Nutraceuticals Product Market: Global Market Size, Segment and Country Analysis and Forecasts (2007-2017),” www.transparencymarketresearch.com/global-nutraceuticals-product-market.html (March 2012).
2. T. Ogura, Y. Furuya and S. Matuura, “HPMC Capsules: An Alternative to Gelatin,” Pharm. Technol. Eur. 10(11), 32–42 (1998).
3. S.M. Ku, et al., “Performance Qualification of a New Hypromellose Capsule: Part I. Comparative Evaluation of Physical, Mechanical and Processability Quality Attributes of Vcaps Plus, Quali-V and Gelatin Capsules,” Int. J. Pharm. 386(1–2), 30–41 (2010).
4. E. Groshens, X. He and D. Cade, “Dissolution Performance in USP SGF Media,” poster presented at the 2009 Annual Meeting and Exposition of the American Association of Pharmaceutical Scientists: www.capsugel.com/media/library/hypromellose-hard-capsules-reproducibility-of-the-in-vitro-dissolution-performance-in-usp-sgf-media.pdf.
