At the recent SupplySide West trade show, nutritional supplement company MitoSynergy celebrated its two newly awarded patents for a Pure Chelation Process (20170274370) and Chlorobis Copper (I) Complex Compositions and Methods of Manufacture and Use (20150224112).
MitoSynergy’s Cunermuspir (copper [I] niacin) is the world’s first bioavailable copper supplement that’s been specially developed to deliver copper in its optimal oxidisation state.
Cunermuspir is created by a process that mimics how plants process copper, allowing bioavailable copper to enter human mitochondria cells and be utilised for the important biochemical process of ATP cycling.
“As the only manufacturer of copper (I) niacin, we take pride in providing manufacturers with a safe form of copper that is bioavailable and can help users by improving energy, increasing mental clarity and reducing general physical discomfort,” said Charlie Barker, CEO of MitoSynergy.
Discussing the product further, he said: “Copper is present in all things animal, vegetable and mineral, and plays an important physiological role in both humans and plants. A disequilibrium with the natural order on Earth results when copper in a form that plants have evolved to process is given to animals. The bioavailability of copper for plants compared with that for humans is not the same.”
“Copper is both toxic but essential,” he adds: “And, for some reason, it’s thought that plant copper is OK for humans to ingest. But, animals have not evolved to utilise copper in its mineral forms. Copper in its mineral form may be toxic to animals that do not have the protein chaperones and other mechanisms to handle it.”
Copper in minerals
Copper is one of the most unique elements on Earth, existing in both a plus one and a plus two oxidative state. Inorganic (mineral) copper is found in nature as a soft metal and in various oxide forms complexed with other elements (such as sulphur and oxygen).
Charlie explains: “Copper comes in three oxidative states: metallic (Earth) copper zero (Cu0) has zero activity, 29 protons and 29 electrons. Earth copper becomes bioavailable to plants by losing two electrons to become cupric copper or Cu2+ (See Figure 1).
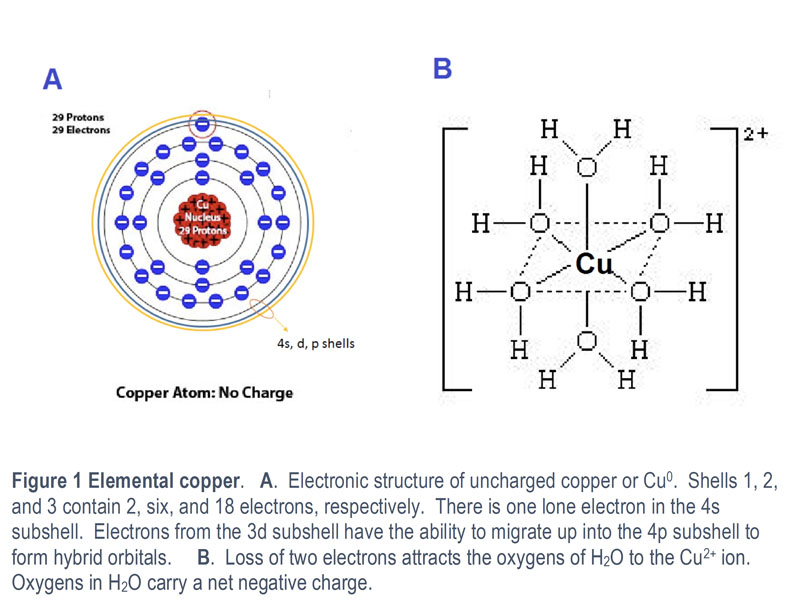
Figure 1: Elemental copper
Some of 3d electrons can be transferred to the 4s and 4p shells to form hybrid bonding orbitals (Figure 1B). Copper in its cupric state (Cu2+), can be usually found bound to water ligands as a co-ordination complex in aqueous solutions (figure 1B).
This micronutrient state is used by plants in soil. Yet, for more than 100 years, modern agricultural practices have been depleting our soils of copper. As a result, all of our plants are mineral deficient before we consume them.1
Copper uptake in plants
Once the Cu2+ is in the plant, it is reduced to Cu+ and chelated. “I believe that this is at the point at which Cu2+ copper becomes Cu+ and, as I like to say, copper for humans,” notes Charlie.
“Other than microbial symbionts, plants use two recognised strategies to absorb copper from soil-bound minerals. Strategy 1 is used by dicots and non-grass monocots; strategy 2 is used by grasses.”
Strategy 1 starts by acidifying the soil around the root using a protein-mediated proton pump.2 Mineral complexed copper is liberated as the Cu2+ cation, which is reduced to Cu+ by a root surface reductase, Fro4/5, and transported via a protein ion channel (COPT1). Strategy 2, the absorption of mineralised copper (and iron), is initiated by the secretion of chelators called phytosiderophores into the rhizosphere.3
Copper uptake in animals
Says Charlie: “Unlike plants, there is scant documentation for a membrane-bound Cu2+ reductase such as Fro4/5 in humans and I believe that the assumption that animals also have a Cu2+ reductase is one of the biggest misconceptions held by science and medicine regarding copper handling."
"Like COPT1 in plants, animals have a Cu+ ion channel called Ctr1. They can also transport Cu2+ via a divalent cation transporter called DMT1. But, I believe, Cu2+ cannot go to where it is supposed to be because it is in the wrong oxidation state and on the wrong side of the membrane!”
In the dietary supplement industry, it’s thought that Cu2+ supplements will interfere with Zn2+ absorption. Ironically, Arredondo et al. demonstrated that DMT1 transported Cu+ ten times better than Cu2+.4
Another potential pathway of copper absorption in animals is the complexation with peptides found in plant and animal protein digests. These are numerous, as copper is an essential cofactor in many plant and animal enzymes.
“If these transporters can be exploited for pharmaceutical purposes, surely they can transport complexed copper,” suggests Charlie: “Hypothetically, copper could bind to the natural dietary substrates of these enzymes and be transported as bound complexes.”
Cuprous and cupric deficient
“Most of us in North America are copper deficient (starving for human proper copper) and, at the same time, we are all copper toxic (accumulating plant copper).
If mineral copper is depleted from the soil, plants and the animals that eat them will, in turn, be copper deficient. Plus, I believe copper deficiency in animals is exponentially compounded when the animal is supplemented with copper in its mineral/plant form,” states Charlie.
He adds: “I believe plant/mineral copper accumulates in our brains. Although there is conjecture in the scientific community, Cu2+ copper may be more prone to accumulate in deleterious deposits such as amyloid plaques in the brains of Alzheimer’s patients … and I’m convinced we make Alzheimer’s disease and other conditions incredibly worse by giving plant copper to humans.”5,6

“I believe that the liver is another site of extracellular copper deposition. Sparks et al. addressed the interplay between the liver detoxification of blood Aß peptides and extracellular Aß amyloid structures in the brain. The addition of only 0.12 ppm of copper (one tenth the Environmental Protection Agency Human consumption limits) to distilled drinking water was sufficient to precipitate the accumulation of Aß in the brains of cholesterol-fed rabbits."
Plus, Sarkar and Roberts examined the role of the Cu2+ specific binding protein, COMMD1, in copper export. One might speculate that if the amount of copper accumulates beyond the ability of COMMD1 and any associated proteins to handle it, copper will accumulate in the liver of humans (as it did in test animals with COMMD1 mutations).”7,8
“As long as COMMD1 is functional and not pushed beyond capacity, all excess plant copper should be conjugated and excreted to bile salts. If these conjugates make it to the colon, though, the Cu2+ has the potential to impact the colonic microflora, which could result in pandemic bacterial imbalances."
"The gut-brain axis is a topic of interest at autismone.org and a great deal of recent peer-reviewed literature. The NIH considers this interaction to be important and has released a Funding Opportunity Announcement to solicit proposals from small businesses and academic research laboratories to better understand this interaction,” comments Charlie.
In summary
“Because mineral/plant copper accumulates in humans, all the copper — other than Cunermuspir (cuprous nicotinic acid) — in dietary supplements, processed food and drinking water will accumulate in our extracellular matrix … loving the brain and liver first."
"It is my opinion that only the cuprous form should be consumed when taking copper supplements,” expounds Charlie, adding: “There are many therapeutic benefits that proper copper in dietary mineral supplementation can be made available to humans, livestock and performance animals.”
“My team and I have developed the world’s first bioavailable copper for humans but will we need everyone's help in joining the cause to educate and correct this misconception about copper and stop us all from being copper toxic and copper deficient at the same time.”
References
- M.S. Fan, et al., “Evidence of Decreasing Mineral Density in Wheat Grain Over the Last 160 Years,” J. Trace Elem. Med. Biol. 22(4), 315–324 (2008).
- A.T. Fuglsang, et al., “Receptor Kinase-Mediated Control of Primary Active Proton Pumping at the Plasma Membrane,” Plant J. 80(6), 951–964 (2014).
- R.D. Graham and J.C. Stangoulis, “Trace Element Uptake and Distribution in Plants,” J. Nutr. 133(Suppl.), 1502S–1505S (2003).
- M. Arredondo, et al., “DMT1: A Physiologically Relevant Apical Cu1+ Transporter of Intestinal Cells,” Am. J. Physiol. Cell Physiol. 284(6), C1525–1530 (2003).
- G.J. Brewer, “Copper-2 Ingestion, Plus Increased Meat Eating Leading to Increased Copper Absorption, Are Major Factors Behind the Current Epidemic of Alzheimer's Disease,” Nutrients 7(12), 10053–10064 (2015).
- W.A. Gunderson, et al., “Local Structure and Global Patterning of Cu2+ Binding in Fibrillar Amyloid-ß [Aß(1–40)] Protein,” J. Am. Chem. Soc. 134(44), 18330–18337 (2012).
- D.L. Sparks, “Cholesterol Metabolism and Brain Amyloidosis: Evidence for a Role of Copper in the Clearance of Abeta Through the Liver,” Curr. Alzheimer Res. 4(2), 165–169 (2007).
- B. Sarkar and E.A. Roberts, “The Puzzle Posed by COMMD1, a Newly Discovered Protein binding Cu(II),” Metallomics 3(1), 20–27 (2011).
