Whether it’s the food they eat, the gym they attend or the fitness activities they participate in, today’s consumers have access to a growing range of options designed to meet their personal preferences and fit into their individual lifestyles.
Yet, when it comes to taking food supplements, most products are offered as traditional tablets and capsules — dosage forms that many people dislike as they can be difficult to swallow and, in some cases, have an unpleasant taste or odour.
To meet the needs and preferences of modern patients and consumers, it is vital that healthcare companies ensure their products are easy to swallow and pleasant to take. What’s more, as convenience becomes key, it’s becoming increasingly important that they are also safe, quick and easy to prepare … and integrate seamlessly into their customers’ daily routines.
Wouldn’t it be great if taking food supplements was as convenient and enjoyable as drinking a cup of coffee?
A new approach to taking food supplements
Fortunately, the latest innovations in dosage form design are helping consumers do exactly that — and HERMES PHARMA is one company that has pushed the boundaries with the introduction of a convenient and innovative dosage form.
With a long history in developing and manufacturing user-friendly delivery systems, the company has this year introduced the HERMES NutriCap, which allows nutraceuticals and food supplements to be prepared at home using Nespresso coffee machines and consumed as an enjoyable hot drink.
As such, HERMES NutriCaps provide additional use for the coffee machines that are already in consumer homes. By using the same preparation method as coffee pods, they are quick and easy to use. Moreover, as HERMES NutriCaps eliminate the inconvenience of having to boil water using a kettle, they are safe and minimise the risk of scalding oneself with boiling water.
Like other user-friendly dosage forms, such as effervescent tablets and instant drinks, HERMES NutriCaps can be designed to come in a range of different flavours to better suit individual consumer preferences. This is in contrast with traditional tablets and capsules, which often do not offer an enjoyable experience.
For pharmaceutical and nutraceutical companies, this flexibility to cater to user desires allows the expansion of product ranges with new dosage forms and additional applications.
Design considerations when developing supplements
There are several design criteria that must be taken into account when developing powders for HERMES NutriCaps compared with traditional tablets and capsules. However, with the right expertise, technologies and experience, these important considerations can be easily navigated, as HERMES PHARMA recently demonstrated with the development of a cough and cold formulation containing vitamin C, zinc and bioflavonoids.
One of the most important factors to consider when manufacturing HERMES NutriCaps is making sure that each dose contains the same amount of nutrients and other ingredients. This is not only essential to ensure consumers receive the correct and safe quantity of nutrient every time, but also to maintain a consistent experience.
Unlike filling hard pods with coffee, which involves using grains of a single material with a relatively uniform size, the production of nutraceutical products is more challenging.
These products are more complex, as they often contain multiple nutrients and excipients with different particle sizes and flow characteristics. As a result, advanced mixing, blending and pod-filling technologies are used to enhance the processability of the ingredients and deliver a consistent dose.
Another critical issue is the need to avoid the risk of carry-over effects that could potentially alter the taste of coffee brewed after preparing a HERMES NutriCap or vice versa. If the nutrients or flavoring excipients do not fully dissolve before the brewing process of the coffee machine is finished, liquid residues may remain in the machine and be transferred to subsequent drinks.
When developing HERMES NutriCap products, it is therefore vital to select fast-dissolving ingredients that can be formulated with a uniform particle size to eliminate the potential for any carry-over effects. In a study of more than 200 customers who were asked to evaluate the taste of coffee brewed after preparing a HERMES NutriCap using their home coffee machine, 84% of those questioned felt that no carry-over effects could be detected (Figure 1).
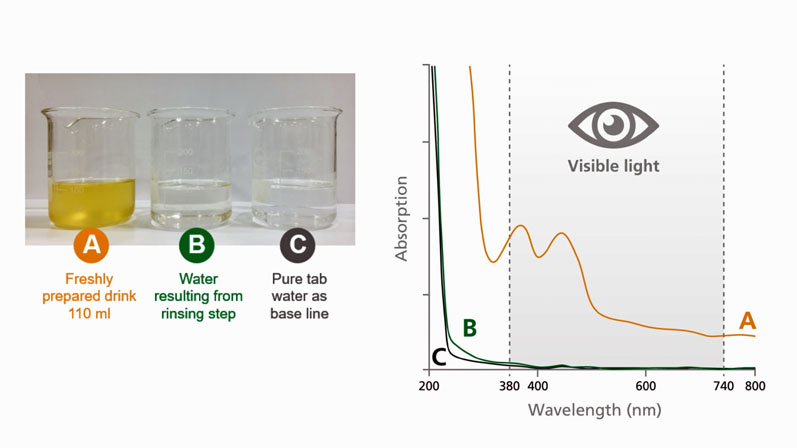
Figure 1: The UV/vis spectrum for the freshly prepared drink (A) shows a high absorption across a wide wavelength range. The spectrum for the subsequent rinsing step (B) shows only minimal to no absorption and is nearly identical to the spectrum for pure tap water (C). This demonstrates that there are no discernible carry-over effects when brewing a coffee directly after a HERMES NutriCap in the same machine
Additionally, as HERMES NutriCap products are consumed as hot drinks, factors such as taste, smell and appearance are more important to consider than when developing traditional tablets and capsules. Many nutrients are bitter, sour, sulphuric, astringent or salty, requiring the careful selection of tastemasking and flavouring excipients to deliver a pleasant oral experience.
Choosing the right flavourings to enhance the taste of nutraceutical products is not always straightforward, and evaluating the outcome of tastemasking efforts can be subjective and time-consuming. However, with the right guidance from experienced formulation experts, systematic product development as well as the use of efficient taste-testing strategies, this process can be shortened considerably.
Similarly, the visual appearance of HERMES NutriCap products can also present challenges if appropriate attention is not paid during formulation development. Some nutrients, such as plant extracts, can be less soluble in water, resulting in precipitation when the drink is dispensed into the cup.
Other components, such as protein-containing ingredients, can cause foaming, compromising product appearance. However, by selecting the right formulation technologies that support the complete dissolution of all components and eliminate foaming, products can be developed with an attractive visual appearance.
Furthermore, in-depth formulation expertise is required to ensure that the product remains stable with time, both in the pod and in the prepared drink. In particular, the stability of ingredients when in contact with hot water is a key consideration during formulation development. These concerns apply not only to the nutrients, but also to flavours and the colour-giving components of the prepared drink, which all have the potential to degrade with time.
Case study: developing a cough and cold formulation
When it came to developing a cough and cold formulation containing vitamin C, zinc and bioflavonoids, HERMES PHARMA considered all of the key design factors highlighted here to develop a highly effective and convenient product that consumers would enjoy.
One of the most critical design considerations was the need to maintain the stability of vitamin C at high temperatures, as the product would be prepared and consumed as a hot drink. By applying appropriate excipients to preserve the stability of the product both in the pod and once dispensed in the cup, a precise amount of vitamin C could be ensured in each dose (Figure 2).
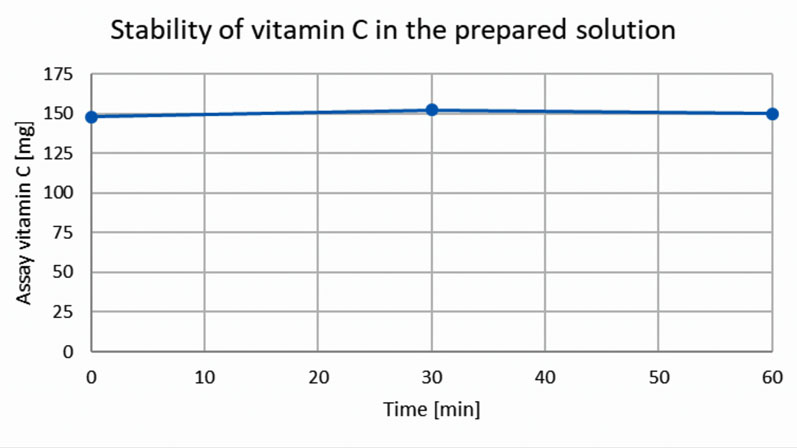
Figure 2: Stability of vitamin C in the prepared solution with time (measured immediately after preparation, after 30 minutes and after 60 minutes), with a starting temperature of 80 °C. The figure shows that the declared content of 150 mg of vitamin C remains stable with time
To further support consistent dosing, it is essential that the contents of the pod dissolve completely. As a result, HERMES PHARMA selected rapidly dissolving ingredients and excipients to support fast, consistent and complete dissolution … well before the preparation of the drink is completed.
Given the low dosage of zinc in the formulation, ensuring content uniformity was particularly important. To deliver a standardised dose, HERMES PHARMA used its pharmaceutical mixing expertise and customised filling equipment — specially adapted from the coffee industry — to meet the needs of handling nutraceutical ingredients and excipients. This ensured that all ingredients were blended uniformly and could be transferred to each HERMES NutriCap in a highly precise manner.
It was also important to avoid the formation of dust during large-scale manufacture of the product, which can occur when processing powders. Dust affects the sealing quality of the pods and thus allows the ingress of humidity. This potentially leads to the formation of lumps of material within the capsule, resulting in inconsistent dosing if the ingredients remain in the NutriCap.
To avoid these issues, HERMES PHARMA used processing and filling technologies that minimised dust formation, ensuring a tight seal and maintaining a consistently high level of product quality and performance.
Conclusion
With consumers increasingly expecting ease and convenience in all aspects of their lives, HERMES NutriCaps are an innovative dosage form that provides nutraceutical developers with the opportunity to extend their product lines and differentiate products from competition.
Although there are a number of additional design issues to consider when developing nutraceutical products as HERMES NutriCaps, when supported with the right knowledge and expertise, healthcare companies can easily leverage this new dosage form to create great-tasting products that consumers will enjoy.





