Osteoporosis is a chronic condition, largely associated with older age groups, in which bones become porous and lose their strength, making them fragile and more susceptible to fracture.1,2
It is widely cited as one of the most common bone diseases and osteoporosis-related fractures affect an estimated one in three women and one in five men older than the age of 50.3
Not only does this disease have a significant impact on an individual’s quality of life, but its consequences impose a growing burden on healthcare systems everywhere.
The management of osteoporosis frequently includes the administration of combined calcium and vitamin D3 (cholecalciferol) oral dosage supplements alongside other treatment regimens.
These supplements must be taken regularly and consistently to be effective. However, the amount of calcium they contain is high, so the tablets are often bulky and have an unpleasant chalky mouthfeel and aftertaste.
These characteristics can make swallowing difficult and turn tablet taking into a highly disagreeable experience, discouraging patient compliance.
Formulating more convenient, user-friendly combined calcium and vitamin D3 dosage forms offers a smart solution … but is technically demanding.
The amount and bulk of the calcium compounds in each dose is challenging in itself and contrasts with the extremely small volumes of vitamin D3 present, making content uniformity difficult to achieve.
Coupled with this is the extreme sensitivity of vitamin D3 to both light and oxygen, necessitating its careful and specialised handling.
Osteoporosis and its management
Bone metabolism is a continuous and lifelong process of growth and resorption. Around 5–10% of the human skeleton is replaced each year through this bone remodelling.4
With ageing and in some disease states, bone resorption exceeds formation, resulting in loss of bone and reduced structural integrity. Osteoporosis is the point at which bone mass drops to a level when the risk of fractures increases (Figure 1).
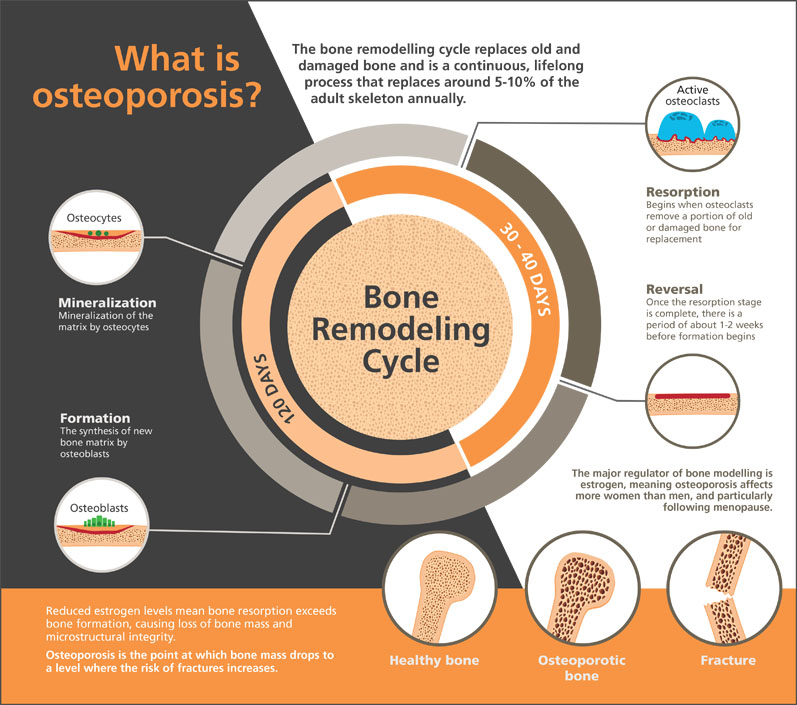
Figure 1: Osteoporosis and the bone remodelling cycle
Osteoporosis affects an estimated 200 million people worldwide and, in a world of ageing populations, it is a growing healthcare challenge.1
Current estimates in the European Union alone are that the fracture-related costs of the disease will increase from €37.5 billion in 2017 to €47.4 billion by 2030.2
Although osteoporosis cannot be cured, an appropriate lifestyle together with medication and dietary supplements can make a difference to the progression and consequences of the disease.
Depending on individual risk profiles, treatment approaches include the use of bisphosphonates, selective oestrogen receptor modulators (SERMs), parathyroid hormone and other hormones as indicated.5
Calcium and vitamin D3 supplementation is widely used — alone or alongside these treatments.6
Calcium is an essential building block in bone metabolism. Its absorption in the small intestine relies on the presence of vitamin D, which the body produces in response to sunlight.
Vitamin D is also present in a small number of foods. However, deficiency is not uncommon, especially in older people or those with restricted exposure to sunlight.
Combined calcium and vitamin D3 dosage forms are an effective and convenient option for supplementation, especially in an older population when many people may already be managing multiple medications.
The need for user-friendly dosage forms
Combined calcium/vitamin D3 supplements are generally taken once per day, and consistent dosing is important for maximum efficacy. However, patients may be discouraged from complying by the bulky nature of the tablets, which can taste unpleasant, feel chalky in the mouth and be difficult to swallow.
As 50% of people across all age groups have difficulties swallowing tablets, this is a significant issue and a problem that tends to increase in the older population.7
Beyond conventional tablets and capsules, a number of different types of more user-friendly formulations are available. Chewable tablets, effervescent tablets, instant drinks and orally disintegrating granules (ODGs) all offer convenient administration.
Each can be formulated to deliver a range of dosages of both calcium and vitamin D3. However, no matter what the formulation or dosage, each necessarily contains large quantities of calcium together with very small amounts of vitamin D3, presenting a variety of formulation challenges.

Understanding the formulation issues
A variety of high-quality, user-friendly dosage forms is required to adequately meet the differing needs of patients who require calcium/vitamin D3 supplementation.
Understanding the formulation challenges associated with combined calcium/vitamin D3 supplements is fundamental to achieving successful products. These are relatively difficult to formulate into user-friendly dosage forms and there are some overarching issues.
As already discussed, the calcium dosage is high, from 500–1200 mg per dose. The only useful, easily available — and economically viable — source of calcium is calcium carbonate, a chemical that is widely used within the pharmaceutical and food industries.
The high molecular weights of alternative organic calcium salts, such as calcium citrate, make them extremely difficult or impossible to formulate as needed.
The greater costs of alternative raw materials would also mean higher retail prices and the risk of excluding certain patient groups on cost grounds from being able to access the supplements.
Although calcium carbonate is the usual calcium source for supplements, its properties are not ideal and present several hurdles that must be overcome during formulation, especially in combined dosages.
Calcium carbonate suffers from poor compressibility, which makes tableting and the creation of an intact, physically stable product difficult; different types of calcium/vitamin D3 formulation require different grades of calcium carbonate, making careful selection important; plus, tablets containing calcium carbonate tend to harden during storage, requiring the inclusion of excipients to mitigate this, especially for chewable tablets.
In contrast, vitamin D3 is present in combined formulations at extremely low concentrations, from 400–2000 IU (1000 international units of pure cholecalciferol per tablet is equivalent to approximately 25 µg).
This disparity between the volume of calcium carbonate and the volume of vitamin D3, especially at higher calcium dosages, means that formulators often struggle with issues of content uniformity and how to ensure consistent dosing.
Also, vitamin D3 is a highly sensitive ingredient that is affected by a number of environmental factors, including exposure to both oxygen and light. It requires controlled handling throughout the product lifecycle to prevent degradation and the use of special additives and excipients to improve its stability.
Unpicking and addressing the challenges
The successful formulation of user-friendly combined dosage forms for calcium/vitamin D3 supplements requires expertise in the dosage forms themselves.
This must sit alongside the ability to understand and manage the challenging characteristics of calcium carbonate and the difficulties of working with minute quantities of highly sensitive vitamin D3.
Calcium carbonate selection
The widespread use of calcium carbonate in pharmaceutical formulations means that many grades are readily available, including light and heavy, with different particle sizes and size distributions.
For the successful formulation of combined calcium/vitamin D3 preparations, it is important to choose calcium carbonate of the right particle size and appropriate granulation properties for the application.
In effervescent drink formulations, for example, the calcium carbonate must be easily dissolvable so that the final product taken by the patient is clear and leaves no residues. However, for chewable tablets and ODGs, when the preparation is not dissolved before consumption, this degree of solubility is irrelevant.
Although all grades of calcium carbonate present problems of compressibility when tableting, light and heavy calcium carbonates differ in their bulk density, which influences both their granulation and compression characteristics. Experience with using different grades for different applications helps to guide informed choices.
Most of the formulation processes for user-friendly dosage forms involve a granulation step, with the addition of vitamin D3 at the subsequent blending stage.
The particle size distributions of the granules and the vitamin D3 are important aspects of controlling the blend uniformity and, therefore, the content uniformity of the final dosages (as described below).

Granulation is a key step in the development and later manufacture of effervescent dosage forms
At HERMES PHARMA, we use TOPO, a proprietary granulation technology that is highly suited to the production of effervescent formulations containing at least one solid crystalline organic acid and at least one alkaline carbonate.
TOPO stabilises these components in air while maintaining solubility in water. In this case, carbonate covers the surface of the citric acid with a slight film to reduce reaction with oxygen.
For dosage forms without a granulation step, such as ODGs, the particle size distribution of the active ingredients and specific excipients must match even more closely to prevent segregation during manufacturing.
The calcium carbonate grade and its granulation properties are critically important, as are particle size analysis and the continued monitoring of all relevant components throughout the formulation and manufacturing processes.
Content uniformity
Dealing with one bulky ingredient and one that is present in only very small quantities means confronting issues of content uniformity to meet pharmacopoeial requirements.
A combined calcium/vitamin D3 chewable tablet, for example, may contain just 20–25 µg of vitamin D3, compared with around 1250–3000 mg of calcium carbonate (equal to 500–1200 mg of calcium) in a tablet that weighs 2–4 g.
Blend homogeneity and content uniformity therefore require considerable attention, while at the same time ensuring that measures are in place throughout the process to minimise vitamin D3 degradation.
There are many points in the process that present a risk to content uniformity. During blending itself, for example, vibration in equipment and on the manufacturing floor is a potential issue to guard against.
Unwanted vibration could result in segregation of the vitamin D3, leading to inconsistencies in the blend reaching the tablet press or sachet filling station.
It is good practice to withdraw samples at many different positions in the powder blender and analyse them to confirm homogenous distribution of the ingredients.
Even when a blend has achieved homogeneity, its uniformity may be threatened during conveyor belt or vacuum hose transport from the bulk container to the tableting press or fill station.
The accumulation of vitamin D3 at various points — in corners or around the edges of processing equipment — can result in doses that are higher or lower than expected.
Overcoming these types of segregation issues requires particle size measurement throughout formulation development, scale-up and into manufacturing.

It is important that the particle size distributions of the different components — the vitamin D3 particles and the granulated materials — are similar to discourage segregation, and that the vitamin D3 particles adhere to the granules without segregating upon vibration.
When undertaking process validation for scale-up into manufacturing, particular attention must be paid to confirming the even distribution of vitamin D3 in the blend.
This involves intensive scrutiny of the first three batches in particular to ensure that they all deliver a uniform product. Once the end product is on the market, a system of ongoing process verification ensures that this quality is maintained.
Vitamin D3 instability
The molecular structure of vitamin D3 is such that it is prone to isomerisation, oxidation and auto-oxidation. Light, humidity and oxygen/air all support these processes; so, throughout formulation, manufacturing and packing, it is necessary to minimise exposure to light and air; environmental humidity must also be kept low.
Manufacturing planning must be optimised to ensure that, once blending is complete, there are no delays in transferring materials to their primary packaging. The use of specialty excipients, including antioxidants, is also needed during manufacture to help reduce oxidative degradation and increase stability.
Ensuring maximum shelf-life
Achieving a long shelf-life is critical for the consistent availability and commercial success of any medication or supplement.
The typical shelf-life of calcium/vitamin D3 combined dosage forms is 24–36 months depending on the type of formulation and is driven by the inherent degradation of vitamin D3.
Protective packaging plays a role in minimising degradation, especially the multidose containers used for products such as effervescent and chewable tablets.
Packaging these tablets in tubes or in foil strips so that each tablet is wrapped individually offers protection, whereas instant drinks and ODGs are packaged in protective sachets.
The tendency of calcium carbonate to harden on storage requires mitigation measures during the formulation of chewable tablets in particular and routine measurement during shelf-life studies.
In conclusion
Osteoporosis is a significant and growing healthcare challenge that typically affects people in older age groups.
Calcium and vitamin D3 supplementation plays an important role in the treatment of this disease and there is increasing demand for combined calcium/vitamin D single dosage forms that are more convenient for the patient to take and aid compliance.
Formulating modern, user-friendly dosage forms, such as effervescent tablets, instant drinks, chewable tablets and ODGs, is challenging when dealing with the high volumes of bulky calcium required and the low volumes of highly sensitive vitamin D3.
Having access to specific knowledge and expertise in these types of formulations helps developers to address the many issues and enables companies to bring these essential products to market more quickly.
References
- http://ow.ly/NGu750CKC92.
- www.bones.nih.gov/health-info/bone/osteoporosis/overview.
- www.osteoporosis.foundation/facts-statistics/epidemiology-of-osteoporosis-and-fragility-fractures.
- www.surgeryjournal.co.uk/article/S0263-9319(14)00225-7/pdf.
- www.osteoporosis.foundation/patients/treatment.
- https://dx.doi.org/10.2147%2Ftcrm.s3552.
- www.swallowingtablets.com.





